Research
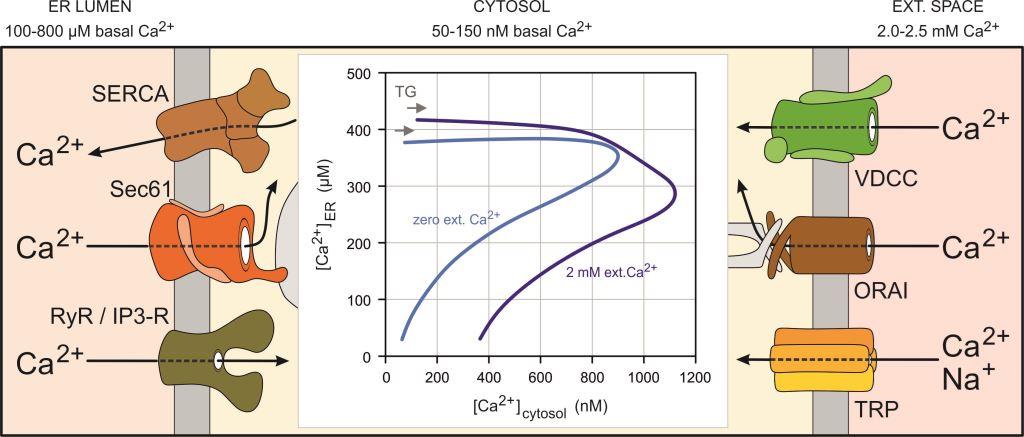
The cytosolic free Ca2+ concentration ([Ca2+]cytosol) is typically 50-150 nM in most mammalian cells, while intracellular Ca2+ stores such as the endoplasmic reticulum (ER) contain free Ca2+ concentrations ([Ca2+]ER) in the range of 100-800 µM at rest. The extracellular Ca2+ concentration is 2.0-2.5 mM in most tissues. Accordingly, the Ca2+ concentration gradient amounts approximately 30000 across the plasma membrane and varies between 666 and 16000 across the ER membrane. Upon stimulation, these Ca2+ concentration gradients determine the Ca2+ fluxes across cell membranes. As illustrated for the effects of thapsigargin (TG), more than a 7-fold increase of cytosolic Ca2+ is produced by the release of small amounts of Ca2+ from ER. Strong clearance mechanisms remove Ca2+ efficiently from cytosol in the absence of external Ca2+ (zero ext. Ca2+) and sustained Ca2+ entry is seen in the presence of external Ca2+ (2 mM Ca2+).
Our research group investigates central aspects of the Ca2+ homeostasis in mammalian cells. Specifically, we study the function and regulation of ion channels localized in the plasma membrane such as voltage-dependent Ca2+ channels (VDCC). ORAI channels that allow the so-called store-operated Ca2+ entry (SOCE) and transient receptor potential (TRP) channels, which can also support Ca2+ entry and modulate the membrane potential, are also in focus of our research projects. Recently, we showed that the translocon, which is formed by Sec61 complexes in the ER membrane, supports Ca2+ fluxes into the cytosol. Other central players in the Ca2+ homeostasis of mammalian cells are ER resident Ca2+ pumps (SERCA) as well as ryanodine and IP3 receptors.
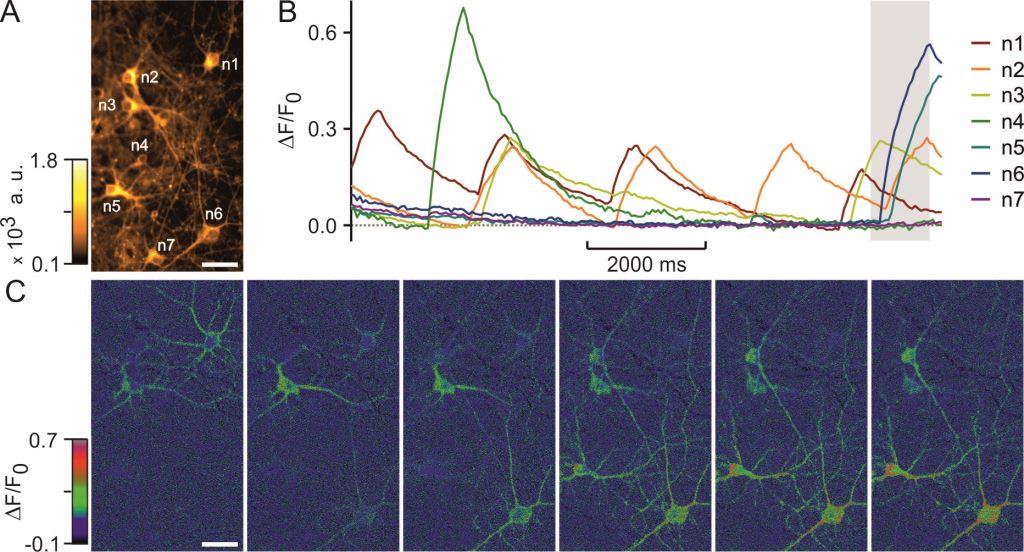
Membrane potential measurements of isolated neurons using a voltage-sensitive dye. Fairless R, Beck A, Kravchenko M, Williams SK, Wissenbach U, Diem R, Cavalié A. PLoS One. 8(3):e58260, 2013
doi: 10.1371/journal.pone
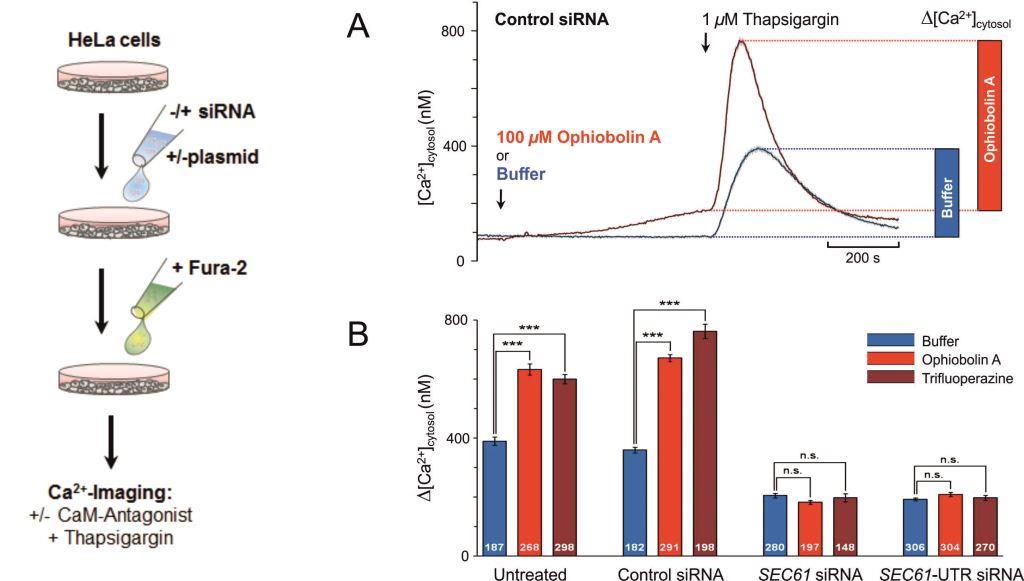
Lang S, Schäuble N, Cavalié A, Zimmermann R. J Vis Exp (53): e2730, 2011
doi: 10.3791/2730.
