Tissue patterning and immune cell modulation in skin
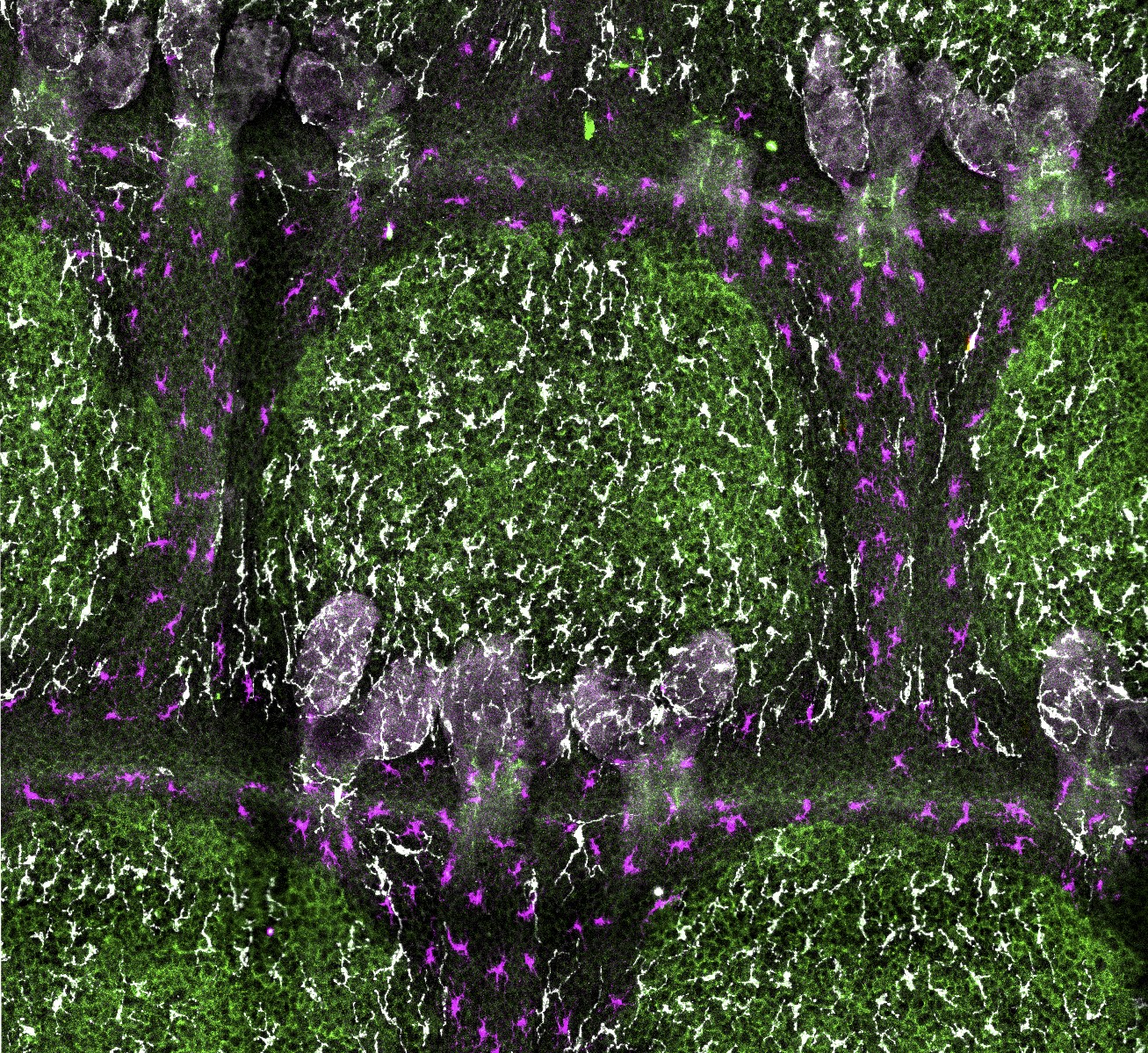
Next to keratinocytes (the main epithelial cell type of the skin), the epidermis hosts melanocytes for skin pigmentation and resident immune cells as first-line host defense. The interdependence of these cell types is currently poorly understood, and the nature, function and molecular basis of underlying heterologous cell-cell interactions are mostly unclear. In murine tail skin, interfollicular epidermis patterns into remarkably orchestrated pigmented ′scale′ and non-pigmented ′interscale′ epidermis. Why and how mature melanocytes confine to scale epidermis is unresolved. We recently delineated a cellular hierarchy among epidermal cell types that determines skin patterning. Already during postnatal development, melanocytes co-segregate with newly forming scale compartments. Intriguingly, this process coincides with partitioning of both Langerhans cells and dendritic epidermal T-cells to interscale epidermis, suggesting functional segregation of pigmentation and immune surveillance. Analysis of non-pigmented mice and of mice lacking melanocytes or resident immune cells revealed that immunocyte patterning is melanocyte- and melanin-independent, and, vice versa, immune cells do not control melanocyte localization. Instead, genetically enforced progressive scale fusion upon Lrig1 deletion showed that melanocytes and immune cells dynamically follow epithelial scale:interscale patterns. Disrupting Wnt-Lef1 function in keratinocytes caused melanocyte mislocalization to interscale epidermis, implicating canonical Wnt signaling in organizing the pigmentation pattern. This work uncovered cellular and molecular principles underlying the compartmentalization of tissue functions in skin (Baess et al., Development 2022).
In addition, we recently investigated molecular players that determine Langerhans cell integrity and networks, see our preprint (Delgado, Burkhart et al., 2025).