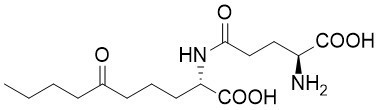
Boletine
γ-Glutamyl boletine is a metabolite of the Japanese mushroom Tylopilus sp. (Boletaceae) showing antibiotic activity.
- R. Watanabe, M. Kita, D. Uemura, Tetrahedron Lett. 2002, 43, 6501–6504.
A straightforward approach towards γ-glutamyl boletine was developed, based on a diastereotopos-differentiating allylic alkylation of chelated amino acid ester enolates. Independent of the configuration of the leaving group in the allylic substrate, the allylation product is obtained as a single stereoisomer. Its configuration is solely controlled by the stereogenic center adjacent to the π-allyl complex formed.
- D. Gawas, U. Kazmaier, “Diastereotopos-differentiating allylic alkylation as key step in natural product synthesis”, Org. Biomol. Chem. 2010, 8, 457–462.