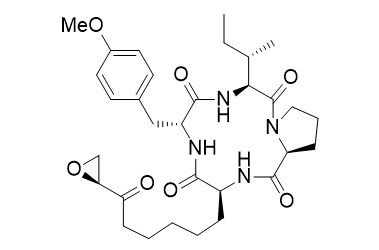
Cyl-1
Cyclic peptides such as Cyl-1, isolated from Cylindrocladium scoparium, are of special interest due to their higher affinity and activity towards Histon-Deacetylases (HDAC). The hydrocarbon chain of the unusual amino acid Aoe, present in this type of HDAC-inhibitors, serves as linker, allowing the zinc-binding motif to coordinate to the Zn2+-ion within the active site.
- A. Hirota, A. Suzuki, H. Suzuki, S. Tamura, Agric. Biol. Chem. 1973, 37, 643–647
- S. Takayama, A. Isogai, M. Nakata, A. Suzuki, Agric. Biol. Chem. 1984, 48, 839–842.
The key step of the synthesis, a chelate enolate Claisen rearrangement, is well suited to generate the unusual amino acid Aoe in enantiomerically pure form.
- P. Servatius, U. Kazmaier, "Total synthesis of the natural HDAC inhibitor Cyl-1", Org. Biomol. Chem. 2018, 16, 3464–3472.
- P. Servatius, U. Kazmaier, "Synthesis and late stage modifications of Cyl-derivatives", Beilstein J. Org. Chem. 2022, 18, 174–181. DOI: 10.3762/bjoc.18.19.