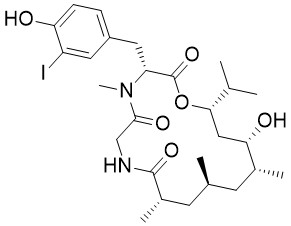
Doliculide
Doliculide was isolated from the sea hare dolabella auricularia, but probably doliculide is not produced by the nudibranch itself, but by the cyanobacteria it shelters or which it feeds. Comparable to the Miuraenamides and Chondramides, Doliculide shows cytotoxicities in the low nM range towards a variety of tumor cell lines. Most effects can be explained by stabilization of the actin skeleton, altering e.g. anaphase chromosome movement which finally leads to apoptosis.
- H. Ishiwata, T. Nemoto, M. Ojika, K. Yamada, J. Org. Chem. 1994, 59, 4710−4711.
- H. Ishiwata, H. Sone, H. Kigoshi, K. Yamada, J. Org. Chem. 1994, 59, 4712−4713.
Doliculide belongs to a group of marine cyclodepsipeptides with interesting biological properties. Apart from a halogenated dipeptide, a polyketide fragment containing 5 stereogenic centers is the most eye-catching element. This building block can be synthesized in a highly stereoselective fashion using only one key reaction: the Matteson homologation. This straightforward protocol allows the introduction of a wide range of substituents at almost any position of a growing carbon chain and is therefore perfectly suited for the synthesis of derivatives for structure-activity relationship studies.
- M. Tost, O. Andler, U. Kazmaier, "A Matteson homologation-based synthesis of doliculide and derivatives", Eur. J. Org. Chem. 2021, 6459–6471. DOI: 10.1002/ejoc.202101345.
- M. Tost, U. Kazmaier, "Synthesis and Late-Stage Modification of (–)‑Doliculide Derivatives Using Matteson's Homologation Approach", Mar. Drugs 2024, 22, 165. DOI: 10.3390/md22040165.