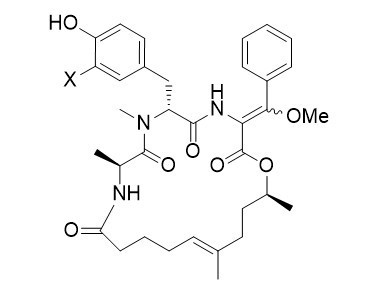
Miuraenamides
From soil samples of japanese halophilic myxobacterium of the species Paraliomyxa miuraensis SMH-27-4 miuraenamides were isolated, cyclodepsipeptides closely related to the jasplakinolides and chondramides. The miuraenamides show antimicrobial activity and inhibit NADH-oxidase. First studies also indicate, that the miuraenamides also stabilize the actin filaments.
- T. Iizuka, R. Fudou, Y. Jojima, S. Ogawa, S. Yamanaka, Y. Inukai, M. Ojika, J. Antibiot. 2006, 59, 385–391.
- M. Ojika, Y, Inukai, Y. Kito, M. Hirata, T. Iizuka, R. Fudou, Chem. Asian J. 2008, 3, 126–133.
- E. Sumiya, H. Shimogawa, H. Sasaki, M. Tsutsumi, K. Yoshita, M. Ojika, K. Suenaga, M. Uesugi, ACS Chem. Biol. 2011, 6, 425–431.
The miuraenamides can easily be obtained by the flexible concept of peptide modification. A combination of aldol reaction/oxidation/methylation on a glycine subunit allows the incorporation of the unusual α,β-unsaturated dehydro amino acid in one of the last steps of the synthesis.
- L. Karmann, K. Schulz, J. Herrmann, R. Müller, U. Kazmaier, "Total Syntheses and Biological Evaluation of Miuraenamides", Angew. Chem. 2015, 127, 4585–4590; Angew. Chem. Int. Ed. 2015, 54, 4502–4507.
- F. A. Gegenfurtner, T. Zisis, N. Al Danaf, W. Schrimpf, Z. Kliesmete, C. Ziegenhain, W. Enard, U. Kazmaier, D. C. Lamb, A. M. Vollmar, S. Zahler, "Transcriptional effects of actin binding compounds: the cytoplasm sets the tone", Cell. Mol. Life Sci. 2018, 75, 4539–4555.
- S. Kappler, L. Karmann, C. Prudel, J. Herrmann, G. Caddeu, R. Müller, A. M. Vollmar, S. Zahler, U. Kazmaier, "Synthesis and biological evaluation of (modified) miuraenamides", Eur. J.Org. Chem. 2018, 6952–6965.
- S. Wang, A. Crevenna, I. Ugur, A. Marion, I. Antes, U. Kazmaier, M. Hoyer, D. Lamb, F. Gegenfurtner, Z. Kliesmete, C. Ziegenhain, W. Enard, A. Vollmar, S. Zahler, "Actin stabilizing compounds show specific biological effects due to their binding mode", Sci. Rep. 2019, 9, 9731.
- S. Kappler, A. Siebert, U. Kazmaier, "Synthesis of New Cyclopeptide Analogues of the Miuraenamides", Curr. Org. Synth. 2021, 18, 418–424. DOI: 10.2174/1570179418666210113161550.
- S. Kappler, U. Kazmaier, "Synthesis of modified miuraenamides – the Ugi approach", Arkivoc 2021, IV, 280–296. DOI: 10.24820/ark.5550190.p011.186.
- S. Wang, M. Meixner, L. Yu, L. Karmann, U. Kazmaier, A. M. Vollmar, I. Antes, S. Zahler, "Turning the Actin Nucleating Compound Miuraenamide into Nucleation Inhibitors", ACS Omega 2021, 6, 22165–22172. DOI: 10.1021/acsomega.1c02838.
- C. Baltes, D. Thalla, U. Kazmaier, F. Lautenschläger, “Stabilizing actin inhibits single cell migration”, Front. Cell Dev. Biol.2022, 10931880. DOI: 10.3389/fcell.2022.931880.