Sex-specific hearing loss
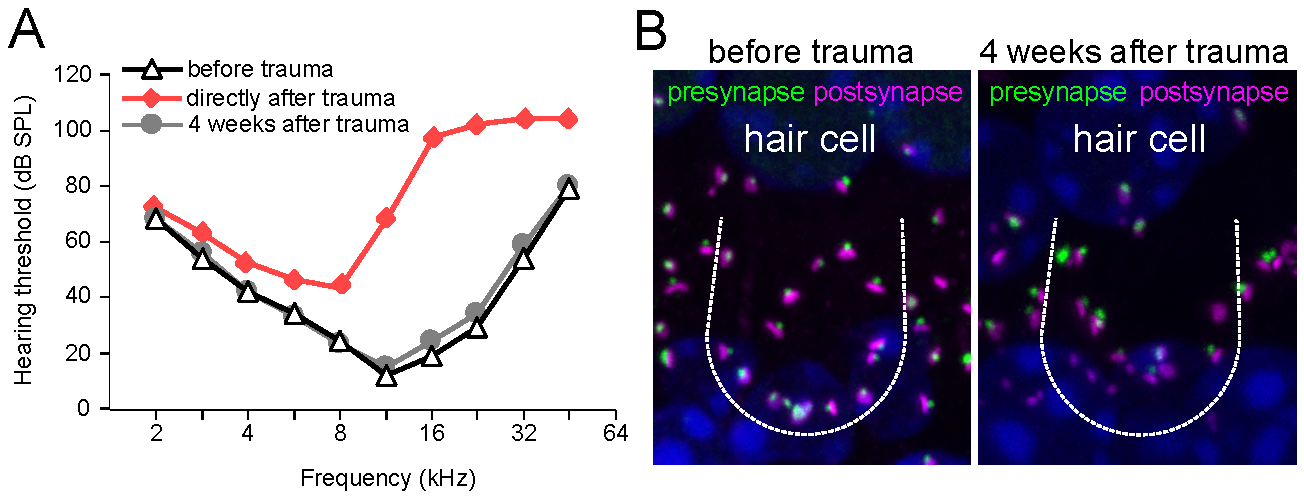
Sensorineural hearing loss caused by malfunction of cochlear hair cells or spiral ganglion neurons is the most common form of hearing loss in humans. It is caused by mutations, aging, toxins, and noise. In both humans and mice, male sex is a risk factor for more pronounced age-related and noise-induced hearing loss.
We will analyze sex-specific consequences of mild and moderate noise exposure on mice with respect to hearing thresholds and the fate of synapses between sensory inner hair cells and primary auditory neurons („cochlear synaptopathy“). Another field of interest is whether sex-specific differences exist in the neuronal processing of auditory signals along the auditory pathway.

Project lead
Prof. Dr. rer. nat. Jutta Engel
Saarland University
Biophysics

PD Dr. rer. nat. Simone Kurt
Saarland University
Center for Integrative Physiology and Molecular Medicine (CIPMM)
Important publications
- Bracic G, Hegmann K, Engel J, Kurt S** (2022). Impaired subcortical processing of amplitude-modulated tones in mice deficient for Cacna2d3, a risk gene for autism spectrum disorders in humans. ENEURO 9, 1-13
- Eckrich S, Hecker D, Sorg K, Blum K, Fischer K, Münkner S, Wenzel G, Schick B, Engel J** (2019). Cochlea-specific deletion of Cav1.3 calcium channels arrests inner hair cell differentiation and unravels pitfalls of conditional mouse models. Front Cell Neurosci 13, 225
- Sonntag M, Blosa M, Schmidt S, Reimann K, Blum K, Eckrich T, Seeger G, Hecker D, Schick B, Arendt T, Engel J, Morawski M (2018). Synaptic coupling of inner ear sensory cells is controlled by brevican-based extracellular matrix baskets resembling perineuronal nets. BMC Biol 16, 99
- Fell B, Eckrich S, Blum K, Eckrich T, Hecker D, Obermair GJ, Münkner S, Flockerzi V, Schick B, Engel J** (2016). α2δ2 controls the function and trans-synaptic coupling of Cav1.3 channels in mouse inner hair cells and is essential for normal hearing. J Neurosci 36, 11024–11036
- Pirone A*, Kurt S*, Zuccotti A, Rüttiger L, Pilz P, Brown D, Franz C, Schweizer M, Rust MB, Rübsamen R, Friauf E, Knipper M, Engel J** (2014). α2δ3 is essential for normal structure and function of auditory nerve synapses and is a novel candidate for auditory processing disorders. J Neurosci 34, 434-445
- Kurt S**, Sausbier M, Rüttiger R, Brandt N, Moeller CK, Kindler J, Sausbier U, Zimmermann U, Winter H, Neuhuber W, Engel J**, Knipper M, Ruth P, Schulze H (2012b). Critical role for cochlear hair cell BK channels for coding the temporal structure and dynamic range of auditory information for central auditory processing. FASEB J 26, 3834-3843
- Joachimsthaler B, Uhlmann M, Miller F, Ehret G, Kurt S**(2014). Quantitative analysis of neuronal response properties in primary and higher-order auditory cortical fields of awake house mice (Mus musculus). Eur J Neurosci, 39, 904-918
- Jaumann M, Dettling J, Gubelt M, Zimmermann U, Gerling A, Paquet-Durand F, Feil S, Wolpert S, Franz C, Varakina K, Xiong H, Brandt N, Kuhn S, Geisler HS, Rohbock K, Ruth P, Schlossmann J, Hütter J, Sandner P, Feil R, Engel J, Knipper M, Rüttiger L (2012). cGMP-Prkg1 signaling and Pde5 inhibition shelter cochlear hair cells and hearing function. Nat Med 18, 252–259
- Baig SM, Koschak A, Lieb A, Gebhart M, Dafinger C, Nurnberg G, Ali A, Ahmad I, Sinnegger-Brauns MJ, Brandt N, Engel J, Mangoni ME, Farooq M, Khan HU, Nurnberg P, Striessnig J, Bolz HJ (2011). Loss of Cav1.3 (CACNA1D) function in a human channelopathy with bradycardia and congenital deafness. Nat Neurosci 14, 77–84
- Kurt S** & Ehret G (2010). Auditory discrimination learning and knowledge transfer in mice depends on task difficulty. Proc Natl Acad Sci U S A 107, 8481-8485
First author *
Corresponding author **